|
This is important for increasing the discovery potential of precision atomic searches for new physics, in particular for atomic parity violation in cesium. These results enable cesium atomic wave functions to be tested in the nuclear region at an unprecedented 0.2% level, and are needed for the development of precision atomic many-body methods. We also deduce accurate values for the BW effect in other isotopes and states of cesium. The effect differs by 0.5% in the hyperfine structure from the value found using the uniform magnetization distribution, which has been commonly employed in the precision heavy-atom community over the last several decades. This value is found from historical muonic-atom measurements in combination with our muonic-atom and atomic many-body calculations. The lines in the sodium lamp are broadened by collisions. Due to the very different emission spectra of these elements, they emit light of different colors. Many street lights use bulbs that contain sodium or mercury vapor. 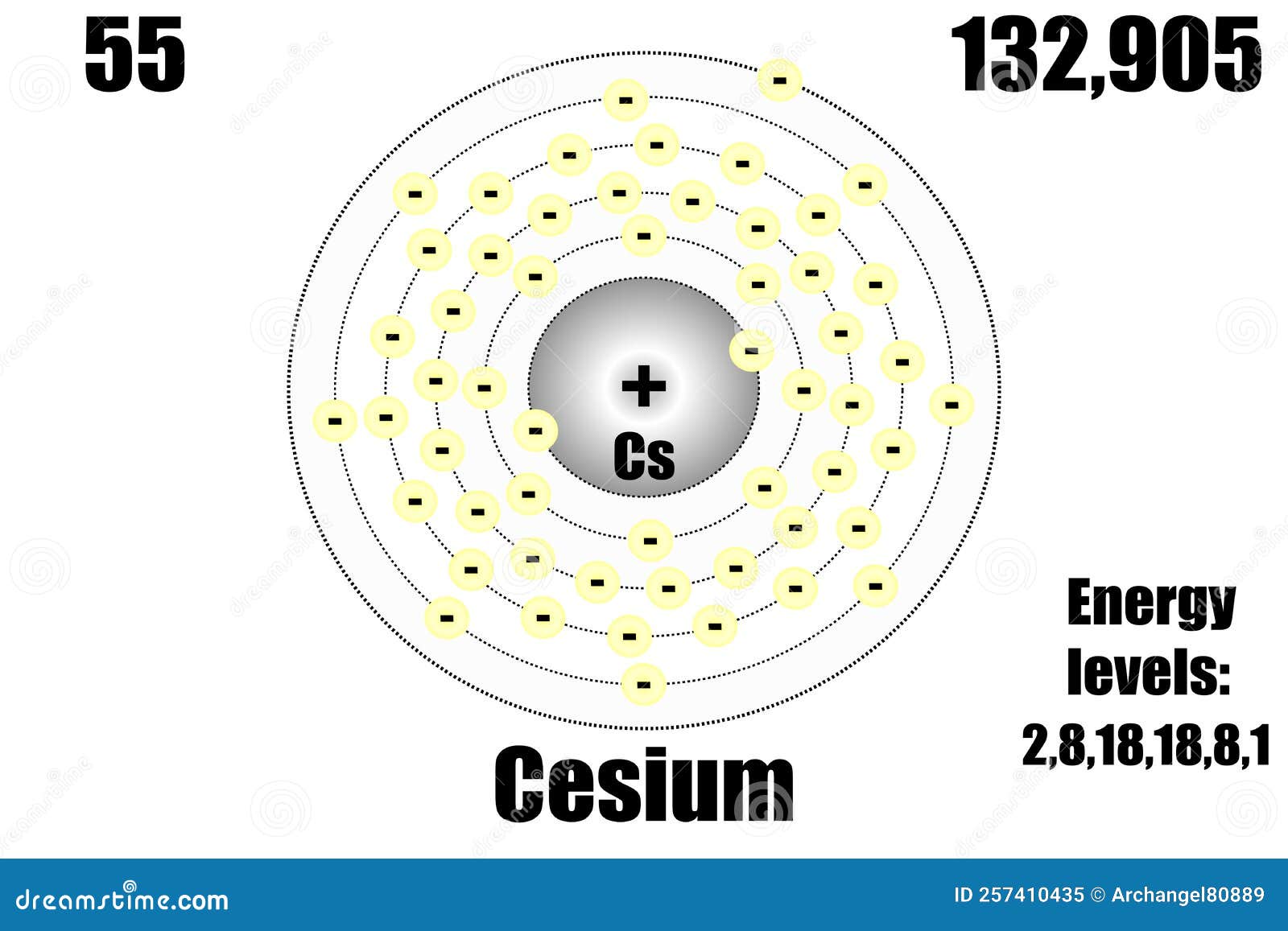
We have obtained an empirical value of − 0.24 ( 18 ) % for this effect in the ground and excited s states of atomic Cs 133. In 1913, a Danish physicist, Niels Bohr (18851962 Nobel Prize in Physics, 1922), proposed a theoretical model for the hydrogen atom that explained its emission spectrum. 2: The emission spectra of sodium and mercury. The Bohr Model has an atom consisting of a small, positively charged nucleus orbited by negatively charged electrons. The finite distribution of the nuclear magnetic moment across the nucleus gives a contribution to the hyperfine structure known as the Bohr-Weisskopf (BW) effect.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
